Strenuous physical exercise and hematological indices of cardiovascular risk: the exception case of a 93-year-old man running a 100-km distance
Introduction
Recent evidence suggests that some hematological parameters may provide valuable clinical information for estimating the cardiovascular risk, but also for assessing therapeutic effectiveness of hypolipidemic drugs, independently from traditional cardiovascular risk factors such as blood lipids, cardiac troponins and unhealthy lifestyle (1). The red blood cell distribution width (RDW) is an indirect measure of anisocytosis, which can now be automatically reported along with many other hematological parameters by the vast majority of modern laboratory instrumentations (2). A high baseline RDW value, along with increasing values of this index of heterogeneity of erythrocyte volumes, are now considered independent predictors of mortality and morbidity in patients with cardiovascular disease and also in the general population (3). In particular, a very recent study showed that an acute 1% variation of RDW independently predicted 30- and 60-day mortality, so conferring an incremental risk of cardiovascular death and mortality of approximately 3-fold (4). The mean platelet volume (MPV) is another hematological index which has gained wide popularity in the pathogenesis of cardiovascular disease, since its value is directly related to the degree of platelet activation and an increase is thereby associated with enhanced thrombotic risk (5). Notably, the variation of MPV is paradigmatic in patients with thrombotic disorders, since higher values are frequently observed before the onset of the occlusive event, whereas the values substantially decrease afterwards, so reflecting platelet exhaustion and hyporeactivity (6).
Due to the increasing scientific interest around these two hematological indices for stratifying the risk of cardiovascular disease, and the still debated relationship between cardiovascular risk and strenuous exercise, in which the well-known health benefits of an active lifestyle may be reversed by the many metabolic perturbations triggered by extreme physical activity (7,8), we describe here the variation of both RDW and MPV in a 93-year-old man, who successfully completed a 100-km distance run.
Athlete selection and workup
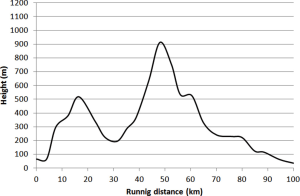
The athlete (WF) was a 93-year-old (body mass index, 17.8 kg/m2; maximum oxygen uptake, 36 mL/min/kg), physically active (about 2.5 h/day of walking) man. The subject has never been an elite or top athlete, has been involved in endurance or ultra endurance competitions since the age of 50 and completed this same event several times before. The event, conventionally known as “100 km del Passatore” (9), consisted of a 100-km run, with a route developing between Firenze and Faenza (Italy) and a total gain of 1,171 m (Figure 1). The 2017 edition was held between the 27th and 28th of May, with official start scheduled at 3 PM of May 27th, in Firenze (Italy). To continuously monitoring his health, the athlete was followed during the entire route by one of the author of this manuscript (CT). The weather conditions were as follows: sunny day, temperature (range), 13–31 °C; humidity (range), 27–72%; wind (mean speed), 7 km/h; partly cloudy with sunny spells.
Blood samples were drawn from an antecubital vein by an expert phlebotomist immediately before the run, after crossing the finishing line, as well as 1, 2 and 7 days afterwards. All samples were collected at approximately the same time of the day (i.e., around 2 PM). The blood was collected in two evacuated blood tubes, the former containing no additives and the latter containing K2EDTA (Vacutest Kima, Padova, Italy). Hematological testing was performed on whole blood with Advia 2120 (Siemens Diagnostic Solutions, Milan, Italy), whereas biochemical testing was performed in serum aliquots using a Roche Cobas 6000 integrated analyzer (Roche Diagnostics GmbH, Mannheim, Germany). In particular, cardiac troponin T (cTnT) was measured with a high sensitivity (HS) immunoassay, characterized by limit of blank and 99th percentile upper reference limit (URL) of 3.0 ng/L and 14.0 ng/L, respectively (10).
Case presentation
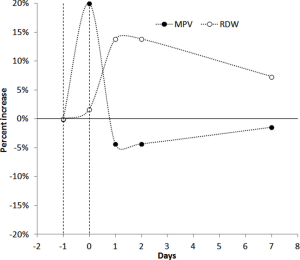
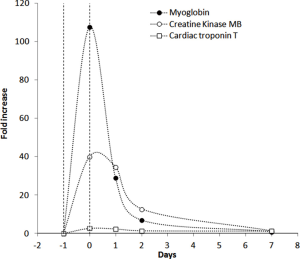
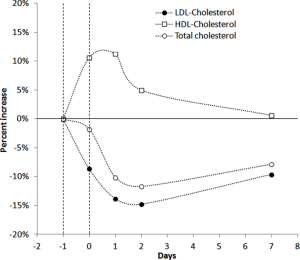
The hematological and biochemical profiles of the 93-year athlete before the start of the event are shown in Table 1. The athlete complete the trial with a finishing time of 17 h 47 min and 41 sec, at an average speed of 5.6 km/h, ending up with a general ranking of 2009 on 2198 regular finishers. The kinetics of RDW and MPV is shown in Figure 2. The RDW value started to increase immediately upon crossing the finishing line and peaked 1 day afterward, displaying a 14% increase from the baseline (i.e., from 12.3% to 14.0%). The RDW then declined to a value still higher than the baseline (i.e., 13.2%) at the 7th day after the trial. The value of MPV exhibited a quite different trend (Figure 2), wherein a consistent increase was observed immediately after crossing the finishing line (i.e., from 7.0 to 8.4 fL), whereas the value then decreased in the following 2 days (i.e., 6.7 fL) and returned to a value nearly identical to the baseline after 1 week. The kinetics of cardiovascular biomarkers is shown in Figure 3. Notably, the serum concentration of cTnT peaked immediately after the end of the run (with a nearly 2.8-fold increase), and then started to decline on the following day, returning to the baseline value after one week. The values of serum myoglobin and creatine kinase MB (CK-MB) also reached the peak after crossing the finishing line (with a nearly 108-fold and 40-fold increase, respectively), and then gradually declined until the 7th day after the run, when their values approximated the baseline serum concentration. The variation of the lipid profile is shown in Figure 4. The value of high-density lipoprotein cholesterol (HDL-C) increased at the end of the run, peaked the day after and then slowly declined during the following days, to approximate the baseline serum concentration after 1 week. The values of both total cholesterol and low-density lipoprotein cholesterol (LDL-C) declined after crossing the finishing line, reached the nadir 2 days after, but 1 week after the trial were still approximately 10% lower than the baseline concentrations.
Table 1
| Parameter | Value | Local reference range |
|---|---|---|
| RDW (%) | 12.3 | 11.5–15.0 |
| MPV (fL) | 7.0 | 7.0–10.0 |
| cTnT (ng/L) | 15.1 | <14.0 |
| CK-MB (ng/mL) | 2.9 | 0–4.9 |
| Myoglobin (ng/L) | 50.0 | 10.0–95.0 |
| Total cholesterol (mmol/L) | 6.2 | <5.2 |
| HDL-C (mmol/L) | 1.6 | <1.0 |
| LDL-C (mmol/L) | 4.1 | <3.4 |
RDW, red blood cell distribution width; MPV, mean platelet volume; cTnT, cardiac troponin T; CK-MB, creatine kinase MB; HDL-C, high-density lipoprotein cholesterol; LDL-C; low-density lipoprotein cholesterol.
Discussion
This article, describing the case of a 93-year-old man who was able to successfully conclude a 100-km run at 5.6 km/h, highlights several important biological aspects. The variations of both RDW and MPV after endurance exercise have been previously described in middle-aged athletes, running much shorter distances. Overall, the kinetics of RDW and MPV was quite similar to that observed in our athlete, both indices displaying an acute increase after the run, with RDW further increasing in the following 24 hours and MPV gradually declining (11). This paradigmatic kinetics is highly suggestive of an acute increase of the cardiovascular risk immediately after endurance exercise, and is also convincingly mirrored by evidence showing that the risk of acute cardiovascular events, also including sudden deaths, is not meaningless during and immediately after medium and long-distance running (12). This has been especially related to development of a transient prothrombotic condition associated with consistent volumes (both in terms of intensity and duration) of aerobic physical exercise (13,14). Unlike the acute risk of cardiovascular events, however, a convincing association has been demonstrated between endurance running, improved quality of life and longevity (12). Overall, the evidence emerged of our case report is in thoughtful agreement with this hypothesis. Despite it seems unquestionable that an immediate risk of cardiovascular events may develop during strenuous exercise, as mirrored by the considerable increase of both RDW and MPV values at the end of the 100-km run, the gradual decline of MPV, lasting for nearly 1 week, is consistent with an ensuing development of platelet hyporeactivity (Figure 2), which may then protect the athlete from the risk of thrombotic events in the days after the long distance run, owing to the well established role of platelets in the pathogenesis of thrombosis (15). The net benefit on cardiovascular risk reduction after the run is also mirrored by the notable variation of HDL-C and LDL-C, with the former parameter consistently increasing for up to 7 days after the run and the latter still remaining 10% lower than the baseline serum concentration 1 week after the end of the run (Figure 4). As regards cardiac troponins, and in agreement with previous studies (16), we also recorded a significant increase (~2.8-fold) immediately after the run in our athlete, but the value rapidly returned to a serum concentration similar to that recorded at the baseline. This kinetics is not consistent with an irreversible cardiomyocyte injury (in which the troponin value typically increases by several order of magnitudes and persists elevated for more than 1 week) (17), but reflects a transitory, reversible and clinically modest cardiac distress (18). Beside general reflections about the variation of some hematological and biochemical parameters in this 93-year-old man, another pragmatic consideration can be done. This old man is an active athlete, without any particular sport qualification, regularly engaged in endurance training. Before the 2017 event of the “100 km del Passatore”, he had already completed many previous editions of this trial. This is another and quite reasonable demonstration that performance of strenuous physical exercise under clinical supervision may be compatible with fitness and longevity, and reaffirms the foremost concept that each laboratory parameter must now be interpreted according to the paradigm of personalized (laboratory) medicine (19).
Conclusions
The paradigmatic variations of some hematological and biochemical parameters in a 93-year-old man who successfully completed a 100-km run suggest that strenuous physical exercise generates considerable changes, triggering an acute prothrombotic state, which is then followed by a longer period of reduced risk of cardiovascular risk.
Acknowledgments
Funding: None.
Footnote
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/aob.2017.07.02). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). Written informed consent was obtained from the athlete for publication of this Case Report and any accompanying images.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Lippi G. Red blood cell distribution width and mean platelet volume: Surrogate markers for, or treatment targets in, dyslipidemia? Clin Biochem 2015;48:555-6. [Crossref] [PubMed]
- Lippi G, Cervellin G, Sanchis-Gomar F. Red blood cell distribution width and cardiovascular disorders. Does it really matter which comes first, the chicken or the egg? Int J Cardiol 2016;206:129-30. [Crossref] [PubMed]
- Salvagno GL, Sanchis-Gomar F, Picanza A, et al. Red blood cell distribution width: A simple parameter with multiple clinical applications. Crit Rev Clin Lab Sci 2015;52:86-105. [Crossref] [PubMed]
- Turcato G, Zorzi E, Prati D, et al. Early in-hospital variation of red blood cell distribution width predicts mortality in patients with acute heart failure. Int J Cardiol 2017; [Epub ahead of print]. [Crossref] [PubMed]
- Lippi G, Mattiuzzi C, Comelli I, et al. Mean platelet volume in patients with ischemic heart disease: meta-analysis of diagnostic studies. Blood Coagul Fibrinolysis 2013;24:216-9. [PubMed]
- Lippi G, Buonocore R, Cervellin G. The Mean Platelet Volume Is Decreased in Patients Diagnosed with Venous Thromboembolism in the Emergency Department. Semin Thromb Hemost 2016;42:632-5. [Crossref] [PubMed]
- Lippi G, Schena F, Guidi GC. Health benefits of physical activity. CMAJ 2006;175:776. [Crossref] [PubMed]
- Kim JH, Baggish AL. Strenuous Exercise and Cardiovascular Disease Outcomes. Curr Atheroscler Rep 2017;19:1. [Crossref] [PubMed]
- LA 100 km del passatore siete vol. Available online: http://www.100kmdelpassatore.it/
- Giannitsis E, Kurz K, Hallermayer K, et al. Analytical validation of a high-sensitivity cardiac troponin T assay. Clin Chem 2010;56:254-61. [Crossref] [PubMed]
- Lippi G, Salvagno GL, Danese E, et al. Variation of red blood cell distribution width and mean platelet volume after moderate endurance exercise. Adv Hematol 2014;2014:192173.
- Day SM, Thompson PD. Cardiac risks associated with marathon running. Sports Health 2010;2:301-6. [PubMed]
- Lippi G, Maffulli N. Biological influence of physical exercise on hemostasis. Semin Thromb Hemost 2009;35:269-76. [Crossref] [PubMed]
- Lippi G, Schena F. Run for Science (R4S): the history of a successful project of precision and laboratory medicine in sport and exercise. J Lab Precis Med 2017;2:11. [Crossref]
- Lippi G, Franchini M, Targher G. Arterial thrombus formation in cardiovascular disease. Nat Rev Cardiol 2011;8:502-12. [Crossref] [PubMed]
- Lippi G, Cervellin G, Schena F. How much myocardium mass may be injured during endurance physical exercise? Clin Chim Acta 2017;470:29-30. [Crossref] [PubMed]
- Cervellin G, Mattiuzzi C, Bovo C, et al. Diagnostic algorithms for acute coronary syndrome-is one better than another? Ann Transl Med 2016;4:193. [Crossref] [PubMed]
- Lippi G, Banfi G. Exercise-related increase of cardiac troponin release in sports: An apparent paradox finally elucidated? Clin Chim Acta 2010;411:610-1. [Crossref] [PubMed]
- Lippi G, Bassi A, Bovo C. The future of laboratory medicine in the era of precision medicine. J Lab Prec Med 2016;1:7. [Crossref]
Cite this article as: Tarperi C, Salvagno GL, Schena F, Lippi G. Strenuous physical exercise and hematological indices of cardiovascular risk: the exception case of a 93-year-old man running a 100-km distance. Ann Blood 2017;2:8.